QMS/cGMP Audit Preparation and Establishment
15,Dec 2019 Mir Shahroz
QMS/cGMP Audit Preparation and Establishment
for Pharmaceutical/Neutraceuticals/Herbal/Medical device/Excipients/Drug Substance company
GAP analysis, System establishment, Facilitation of Regulatory Agency Audit, Deficiency response (Agency) etc,
mail: info@medwisdom.in; Phone: +91-9264127040
GMP & QMS
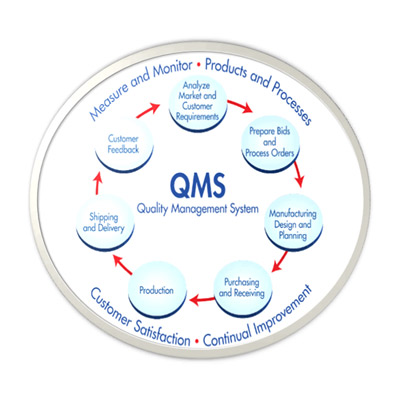
It is a system for ensuring that products are consistently produced and controlled according to quality Standards/Regulations/Directives (WHO, PIC/S, ICH Q11, 21-CFR Part 210/211 (US-FDA), EudraLex - Volume 4-Good Manufacturing Practice (GMP) guidelines (European Commission), etc.
DEFINITION:
As per WHO: It is designed to minimize the risks involved in any pharmaceutical production that cannot be eliminated through testing the final product. The main risks are: unexpected contamination of products, causing damage to health or even death; incorrect labels on containers, which could mean that patients receive the wrong medicine; insufficient or too much active ingredient, resulting in ineffective treatment or adverse effects. GMP covers all aspects of production; from the starting materials, premises and equipment to the training and personal hygiene of staff. Detailed, written procedures are essential for each process that could affect the quality of the finished product. There must be systems to provide documented proof that correct procedures are consistently followed at each step in the manufacturing process - every time a product is made.
It is important to note that cGMPs are minimum requirements. Many pharmaceutical manufacturers are already implementing comprehensive, modern quality systems and risk management approaches that exceed these minimum standards.
References.:
ISO 9001: 2015 (Quality Management system)
ICH Q7 Good Manufacturing practice
ICH Q9 Quality Risk Management
ICH Q10 Pharmaceutical Quality system
European Commission EudraLex - Volume 4 - Good Manufacturing Practice (GMP) guidelines
US-FDA : 21 CFR Part 210/211/820
cGMP
Pre-GMP Inspection for Facility Readiness:
Medwisdom primary objective is to help attain a sustainable state of Permanent Inspection Readiness through the mock audits. To achieve this, Medwisdom ensures a robust and effective Pharmaceutical Quality System is put in place and monitors the effectiveness and suitability through periodic GMP Audit and Quality Management review.
Mock Audits of the Quality Management System helps clients to face regulatory authority inspections confidently. A mock regulatory inspection is an audit conducted by Medwisdom simulating a GMP Audit by a competent Regulatory authority.
Medwisdom works with clients in conducting a GMP Audit for their facilities, systems and / or procedures to support their systems for achieving and maintaining Permanent Inspection Readiness at site supported by advisories on preparations to face any upcoming inspection by any Regulatory Agency.
In response to a client’s needs, Medwisdom experienced team of Auditors will conduct a GMP Audit taking a risk-based approach in accordance with ICH Q9 and Q10. Prior to audit, the team will discuss and formulate the appropriate form for the post-audit report. This report can range from a tabulation of findings to an in-depth analysis of gaps and development of a risk-based program of remediation activities.
Following the audit, Medwisdom also provides further support to implement all of the client’s gap remedial activities till closure of audit observations.
The scope of pre-GMP inspection is as follows:
- Analysis of the current GMP status of a Manufacturing facilities
- Audit report and improvement measures
- Review of GMP documentation prepared by Client’s team
- Building up your quality systems
- Mock inspections and preparation for Agency-inspections
- Facilitation of Agency GMP Inspection (e.g., Initiation/ Trigger of GMP inspection, Facilitation of GMP inspection, Closure of Audit finding)
Vendor Qualification:
- Raw material vendor audits
- Packaging material vendor audits
- Active pharmaceutical ingredient (API)/Drug substance vendor audits
- Excipient vendor audits
- Contract testing laboratories audits
Vendor qualification is important tool for quality management system. The following wording is taken from the GMP standard/Regulation/Directives related to vendor qualification. So Vendor qualification is an essential requirements for Quality management system.
"The selection, qualification, approval and maintenance of suppliers of starting materials, together with their purchase and acceptance, should be documented as part of the pharmaceutical quality system"
"The selection, qualification, approval and maintenance of suppliers of primary and printed packaging materials shall be accorded attention similar to that given to starting materials."
Chapter 7 of the EU-GMP Guidelines (Outsourced Activities) describes the responsibilities of the Contract Giver when it comes to contract manufacturing and testing. He needs to assure the control of the outsourced activities, incorporating quality risk management principles and including continuous reviews of the quality of the Contract Acceptor's performance. Audits are helpful tool to asses the "legality, suitability and the competence of the Contract Acceptor". The new Chapter 7 was obviously designed to intensify the control of Contract Acceptors by the Contract Giver and extend those controls to subcontractors.
ISO 9001:2015:
ISO 9001 is defined as the international standard that specifies requirements for a quality management system (QMS). Organizations use the standard to demonstrate the ability to consistently provide products and services that meet customer and regulatory requirements. It is the most popular standard in the ISO 9000 series and the only standard in the series to which organizations can certify.
ISO 9001 was first published in 1987 by the International Organization for Standardization (ISO), an international agency composed of the national standards bodies of more than 160 countries. The current version of ISO 9001 was released in September 2015.
ISO 9001 helps organizations ensure their customers consistently receive high quality products and services, which in turn brings many benefits, including satisfied customers, management, and employees.
Because ISO 9001 specifies the requirements for an effective quality management system, organizations find that using the standard helps them:
- Organize a quality management system (QMS)
- Create satisfied customers, management, and employees
- Continually improve their processes
- Save costs
ISO 13485: 2016:
ISO 13485:2016 was written to support medical device manufacturers in designing quality management systems that establish and maintain the effectiveness of their processes. It ensures the consistent design, development, production, installation, and delivery of medical devices that are safe for their intended purpose.
TRAINING
Training is an investment in people that pays its dividends in a more skilled workforce, improved productivity, and higher levels of product and service quality.
Training is recognized in the preamble to the current good manufacturing practice regulations as a dynamic process that cannot be satisfied by a single training course to be given to an employee at the time of hire. Training should be a dynamic process, in respect to both, the training an employee receives during his or her career with a firm, and in terms of ensuring that training programmes and materials keep pace with job requirements and performance expectation.
A WHO guide to good manufacturing practice (GMP) requirements – Part 3: Training:







